
(Bilastine)


Drynol 20mg Tablets
Symptomatic treatment of allergic rhino-conjunctivitis (seasonal and perennial) and urticaria
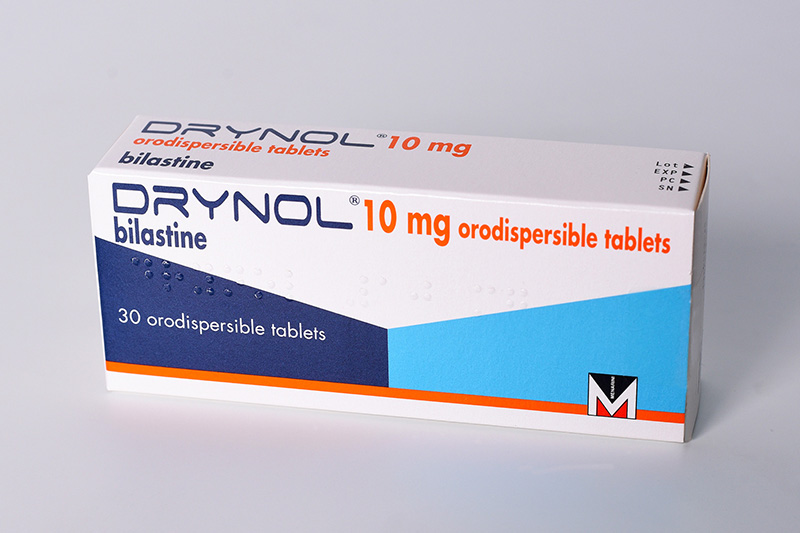
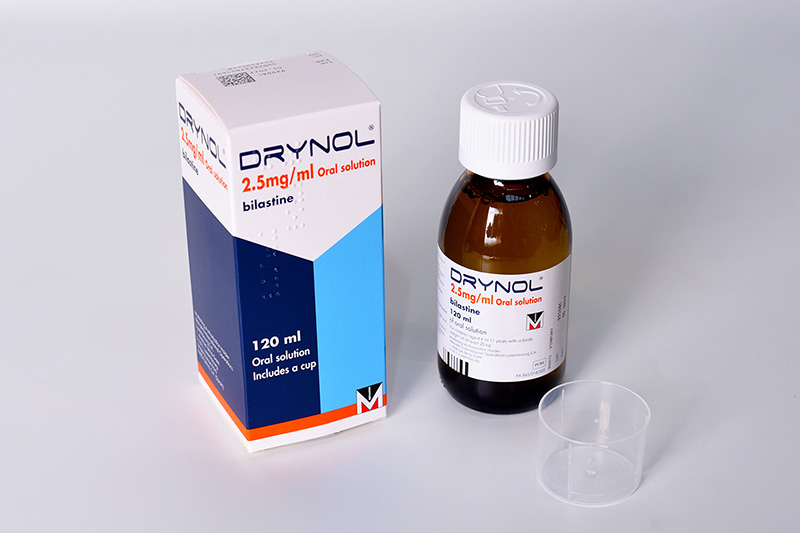
Drynol 10 mg orodispersible tablets and Drynol 2.5mg/ml oral solution
Symptomatic treatment of allergic rhino-conjunctivitis (seasonal and perennial) and urticaria in children aged 2 to 11 years with a body weight of at least 15 kg
Product Information

Abbreviated Prescribing Information
- Drynol 20 mg tablets (bilastine)
- Drynol 10 mg orodispersible tablets and 2.5mg/ml oral solution (bilastine)
Abbreviated Prescribing Information: Drynol 20 mg tablets (bilastine).
Please consult the Summary of Product Characteristics (SmPC) for full prescribing information.
Presentation: Oval biconvex scored white tablets containing 20mg bilastine.
Uses: Symptomatic treatment of allergic rhino-conjunctivitis (seasonal and perennial) and urticaria.
Dosage and administration: Oral administration. Adults and adolescents (12 years of age and over) take 1 tablet once daily. Take one hour before or two hours after food or fruit juice. Elderly, renal impairment or hepatic impairment: no dose adjustment required. Children under 12 years: no information.Duration of treatment: For allergic rhino-conjunctivitis the treatment should be limited to the period of exposure to allergens. For seasonal allergic
rhinitis treatment could be discontinued after the symptoms have resolved and reinitiated upon their reappearance. In perennial allergic rhinitis continued treatment may be proposed to the patients during the allergen exposure periods. For urticaria the duration of treatment depends on the type, duration and course of the complaints.
Contraindications: Hypersensitivity to the active substance or to any of the excipients. Warnings and Precautions: Avoid co-administration with P-glycoprotein inhibitors in patients with moderate or severe renal impairment. Caution should be administering bilastine to patients who are at increased risk of experiencing QT/QTcprolongation.
Interactions: Food, grapefruit juice, ketoconazole, erythromycin, diltiazem.
Pregnancy and lactation: Avoid use during pregnancy. Assess before prescribing in lactating mothers.
Side-effects: Common: somnolence, headache. Uncommon: oral herpes, increased appetite, anxiety, insomnia, dizziness, tinnitus, vertigo, right bundle branch block, sinus arrhythmia, electrocardiogram QT prolonged, other ECG abnormalities, dyspnoea, nasal discomfort, nasal dryness, upper abdominal pain, abdominal pain, nausea, stomach discomfort, diarrhoea, dry mouth, dyspepsia, gastritis, pruritus, fatigue, thirst, improved pre-existing condition, pyrexia, asthenia, increased gamma-glutamyltransferase, alanine aminotransferase increased, aspartate aminotransferase increased, blood creatinine increased, blood tryglicerides increased, increased weight. Frequency not known: palpitations, tachycardia, hypersensitivity reactions and vomiting.Pack size: 30 film-coated tablets.
Legal category: POM. Product Authorisation Numbers: PA 865/18/1.
Product Authorisation Holder: Menarini International Operations Luxemboaurg S.A, 1 Avenue de la Gare, L-1611 Luxembourg
Marketed by: A. Menarini Pharmaceuticals Ireland Ltd. Further information is available on request from A. Menarini Pharmaceuticals Ireland Ltd., 2nd Floor, Castlecourt, Monkstown Farm, Monkstown, Glenageary, Co. Dublin A96 T924 or may be found in the SmPC.
Date of Preparation: April 2025
Abbreviated Prescribing Information: Drynol 10 mg orodispersible tablets and 2.5mg/ml oral solution (bilastine).
Please consult the Summary of Product Characteristics (SmPC) for full prescribing information.
Presentation: Orodispersible tablets (ODT): round, slightly biconvex white tablets containing 10 mg bilastine. Oral Solution (OS): clear, colourless, slightly viscous aqueous solution.Uses: Symptomatic treatment of allergic rhino-conjunctivitis (seasonal and perennial) and urticaria in children aged 2 to 11 years with a body weight of at least 15 kg.Dosage and administration: Oral administration. Children aged 2 to 11 years with a body weight of at least 15 kg. ODT: Take 1 tablet once daily. OS: Take 4 ml of oral solution once daily. ODT and OS: Take one hour before or two hours after food or fruit juice. No dose adjustment required in hepatic or renal impairment. In adults and adolescents (over 12 years of age) the administration of Drynol 20 mg tablets is appropriate.Contraindications: Hypersensitivity to the active substance or to any of the excipients. Warnings and Precautions: Avoid co-administration with P-glycoprotein inhibitors in patients with moderate or severe renal impairment. Do not use in children under 6 years of age. Caution should be exercised when administering bilastine to patients who are at increased risk of experiencing QT/QTc-prolongation. Drynol oral solution contains methyl parahydroxybenzoate (E218) and propyl
parahydroxybenzoate (E216) which may cause allergic reactions (possibly delayed). Interactions: No interaction data available for children. Consider interactions as for adults: food, grapefruit juice, ketoconazole, erythromycin, diltiazem; medicinal products that are substrates or inhibitors of P-gp, such as cyclosporine.
Pregnancy and lactation: Avoid use during pregnancy. .
Side-effects: Common: headache and allergic conjunctivitis. Uncommon: abdominal pain/upper abdominal pain, diarrhoea, nausea, lip swelling, rhinitis, dizziness, loss of consciousness, eye irritation, eczema, urticaria, fatigue. Frequency not known: palpitations, tachycardia, hypersensitivity reactions, and vomiting. Pack size: ODT: 30 orodispersible tablets. OS: One bottle containing 120 ml oral solution.
Legal category: POM. Product Authorisation Numbers: PA 865/18/02 and PA 865/18/03.
Product Authorisation Holder: Menarini International Operations Luxemboaurg S.A, 1 Avenue de la Gare, L-1611 Luxembourg
Marketed by: A. Menarini Pharmaceuticals Ireland Ltd. Further information is available on request from A. Menarini Pharmaceuticals Ireland Ltd., 2nd Floor, Castlecourt, Monkstown Farm, Monkstown, Glenageary, Co. Dublin A96 T924 or may be found in the SmPC.
Date of Preparation: April 2025.
Adverse events should be reported. Healthcare professionals are asked to report any suspected adverse events via: HPRA Pharmacovigilance Website: www.hpra.ie. Adverse events should also be reported to A. Menarini Pharmaceuticals Ireland Ltd. Phone no: 01 284 6744
IR-MEN-34-2025 Date of Preparation: June 2025
